|
Optional Unit
(18d) Work Against an Electric Force:
The Van De Graaff generator
|
Electric Forces Electrophorus expt. |
Many textbook examples on work are based on overcoming gravity--as for instance in preceding section #18. If you lift a brick from the ground to the table, you perform (or "invest") work and gain potential gravitational energy, and this energy can be converted back, to the work of raising a different brick. But nature also has other forces, such as the electric force.
Electric ForcesOrdinary matter consists of negative electrons and positive atomic nuclei, and their electric charges attract each other. That is what holds atoms together! Usual matter has equal amount of positive and negative electric charge.However, by rubbing (say) glass with a dry wool cloth, electrons can be removed, leaving it positive, while the same electrons are picked up by the cloth which therefore gets a negative electric charge. Other materials (like amber) attract electrons from the cloth and get negative when rubbed. Objects with positive charge attract those with negative charge, but two positively charged objects, or two negative ones, repel each other. There exist other ways of detaching electrons from matter, but the process is self-limiting. Suppose you deposit extra electrons on an object: it the becomes negatively charged, strongly repelling any additional electrons and by this not allowing any addition to its charge. Mathematically this can be expressed by a negative "electric pressure" (also known as negative "voltage", since it is measured in units called volts) which pushes back any other negative charges. If that voltage gets too high, electrons may be pushed off as a spark, and a symmetric effect exists with positive charge, which can forcibly pull electrons from surrounding material. That is what happens in lightning, when processes in a thundercloud (see below) separate electric charges and create high voltages.
|
|
In general, sharp points create sparks even at low voltages, while large smooth spheres resist sparking. The interesting thing to note is that the repelling (or attracting) force only exists outside the sphere: inside a hollow enclosure no electric force or voltage can be observed. You may be pleased to know that if you sit inside a closed aluminum airplane cabin when it is struck by lightning, the metal enclosure protects you from any electric effects.
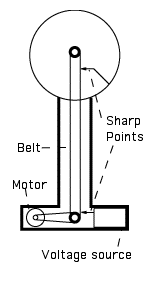
Van de Graaff's InventionIn 1929 Robert Van de Graaff in Princeton found a new way of charging a hollow sphere to high voltages. He mounted it on top of a high insulating column, inside which a rubber belt ran around two pulleys--one inside the sphere, the other (turned by a motor) in the base. Also in the base was a source of negative voltage, attached to a series of sharp points, which sprayed negative electric charge onto the belt. Positive electric charge can also be sprayed--but here it is convenient to talk about electrons. The other side of the voltage source was connected to the ground, so that as electrons were sprayed off, others came up and replaced them, leaving behind no excess positive charge.Since rubber does not conduct electricity (that is, does not allow electric charges to move from one point to another, as a metal would), the belt carried its charges into the sphere, inside which another series of sharp points, placed close to the belt, skimmed them off. Wires connected the points to the sphere, so the charge distributed itself all over it. Gradually, the sphere became more and more negative. That did not prevent the unloading of electrons inside the hollow interior, but the voltage outside, however, could rise to a few million volts. When that voltage became high enough, a lightning-like spark usually leaped to the ground or to some convenient metal target. The Boston Museum of Science has such machines, and uses them to provide visitors with impressive demonstrations.
Energy and Work DoneAs the spheres charge up, they accumulate electric energy. Where does that energy come from? Simple. We said that even when the sphere is charged up to a million volts, inside it electric forces are absent, posing no obstacle to skimming charge off the belt. But outside it, such forces do exist! As the belt carries negative electric charge towards the sphere, it must overcome the repelling force F of the negative charge already on it ... and perform work! The motor must overcome that repulsion, and is therefore the energy source. A student turning the lower pulley by a crank could also provide energy, but should watch out for those sparks!
|

Lightning(I am grateful to Prof. Martin Uman of the U. of Florida in Gainesville for correcting the initial version of this section).Could a similar process be responsible for the high voltages that cause lightning in thunderstorms? A thunderstorm cloud is essentially a violent upward flow of humid air. Rising air expands and cools, but the surrounding air at higher levels is cooler, too: what determines whether a flow continues to rise or not is whether it is warmer or cooler than the air around it. The rising flow in a thunderstorm gives up humidity in the form of rain (cooler air cannot hold as much water) and that process, it may be shown, provides extra heat. The water therefore keeps the air warmer than its surroundings, and it keeps rising. The result is motion in two directions: a wind blows upwards, and meanwhile raindrops fall through it towards the ground. Some raindrops are blown upwards by the wind, to higher levels where they freeze, and this too helps keep the air warmer. (Orange growers in Florida spray water when temperatures drop below freezing: the water freezes and falls to the ground, while orange trees are kept warmer.) Ultimately they may come down again as hail, commonly associated with thunderstorms. Big hailstones apparently start as regular hail, collect more water as they fall, then are blown upwards again. Freezing is important, because observations suggest that thunderstorm electricity is created not by liquid water, but by ice. This might be related to the fact that when water freezes, it gives up "latent heat" of about 80cal/gr associated with the liquid state. That heats the surrounding atmosphere, and might invigorate upward drafts. The ice fragments created in the cloud come in various sizes: while large ones tend to fall down, small slivers are generally blown upwards, and the two kinds collide. The collisions may separate charge (a bit like frictional electricity): the little slivers tend to lose electrons and become positive, and because the total electric charge is zero, those lost electrons give a negative charge to the larger ice particles. The magnitude of the effect depends strongly on temperature--even the sign of the transferred charge reverses for some temperatures--and this leads to additional features of lightning generation. The process is being studied in the lab, by Dr. Clive Saunders of Manchester University and by others. One may speculate that lightning associated with volcanic eruptions may have a similar origin, in encounters between small fragments rising with hot volcanig gases and larger ones, blown upwards but soon falling back. Because the two types of ice fragments have opposite charges, they attract each other: but gravity pulls the bigger ones down, while the wind blows smaller slivers even higher, and in separating the two types, these two forces perform work against the electric attraction. The situation is therefore somewhat similar to Robert Van de Graaff's machine, except that there the rubber band overcomes electric repulsion, while here, the forces of the wind and of gravity overcome an attraction. Still, work is work, and by performing it the process increases the energy stored in the system. The top of the cloud, where the little slivers end up, becomes charged to a high positive voltage, until the air cannot contain the growing electric charge any more, and... FLASH! BOOOOM!
TidbitThe way electric charge is sprayed from a high-voltage source onto the belt of the Van de Graaff generator is also at the heart of the xerographic copier or "xerox machine." In both terms, the prefix "xero-" comes from the Greek word for "dry," implying a printing process using dry ink.In such a copier, electric charge is sprayed onto a rotating roller made of a special insulator, which conducts electricity when white or blue light shines on it. After the roller is charged, the image of the copied page is beamed onto it, and all parts of the image which are white become electrically conducting and lose their charge, while images of black letters stay charged. As the disk rotates further, very fine carbon powder ("dry ink", also containing a little binding glue) is attracted to the electric charge and clings to the roller, and still further that "ink" is transferred to a paper page, to which it bonds with the help of heat. Laser printers operate on similar principles.
Clinging SheetsBesides copying text and images on sheets of paper, the xerographic copier can also make copies on transparent sheets of acetate or some other plastic material, for use in transparency projectors. If you ever produced multiple transparencies like that and stacked them together, you probably found that their electric charge made them cling together rather strongly.You can of course pull apart such sheets, but their electrification is not removed. In fact, it seems worse than before! What happens is that by pulling the sheets apart, you had to perform work against the electric force, and that raised the voltage of the electric charge, just as in thundercloud ice and in the Van de Graaff generator. A similar effects happens when you pull garments made of synthetic fibers, which charge up by friction, out of a clothes dryer. By pulling them apart you perform work and raise their voltage, until sparks can result and even small electric shocks. An old electrostatic device actually exists for elevating static voltages by such means--the "electrophorus" invented in 1782 by Alessandro Volta, the one who later devised the first electric battery ("voltaic pile") and whose name is honored in the unit named volt, measuring what we commonly refer to as "voltage." About those clinging sheets: To be safe, do not stack the sheets as they come out of the copier. Lay them aside individually (on a metal table, shelf or cabinet is best) and let them cool. Afterwards, when you stack them, you may separate them with sheets of paper.
Exploring FurtherA site picturing the Van de Graaff Generators of the Boston Museum of Science. Built by Robert Van de Graaff after he became a professor at MIT, they were later given to the museum. Their story, illustrated by unique photographs, is found here, while the life of Prof. Van de Graaff is described here. A dramatic photograph of the Boston generator in action appeared in the "National Geographic" magazine, issue of October 2001, page 10.The "Van de Graaff page" by the "Science Hobbyist Static Electricity Science Club" http://www.amasci.com/emotor/vdg.html. A home experiment on amplifying a voltage by performing work against the electric force--"Frying Pan Electrophorus." An article on the life of Robert Van de Graaff can be found on p 463-7 in issue 8 of "The Physics Teacher", vol 42, November 2004. Strictly for experts with access to a scientific library: For a look at the wide ranging and complicated evidence on the charging of water droplets and ice fragments in a cloud, see "The Physics of Clouds" by Basil J. Mason, xvi + 671 pp, 2nd edition Oxford 1971.
... And by the way: Van de Graaff's memory was honored by having one of the craters on the far side of the Moon named for him. Later the subsatellites of Apollo 15 and 16 (1971-2) found that the crust of the Moon was magnetized in patches--like what was later found for Mars, though the Moon's field is much weaker. A very intense magnetic patch was found near the Van de Graaff crater.
Questions from Users: Do Cosmic Rays produce lightning? *** Remote sensing of Thunderstorms *** The Path of Lightning *** Charging of Earth by lightning: + or – ? *** Why is lightning jagged? |
Next Stop: #19 Motion in a Circle
Timeline Glossary Back to the Master List
Author and Curator: Dr. David P. Stern
Mail to Dr.Stern: stargaze("at" symbol)phy6.org .
Reformatted 24 March 2006 ;
Updated 8-17-2013 ; and again 13 October 2016